Science & Tech
UN declares International Day of Women in Science
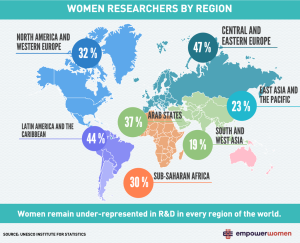
From Roberta Bondar to Harriet Brooks, Canada has more than its fair share of women scientists to be proud of. However women are still a minority in the STEM fields
- 472 words
- 2 minutes
This article is over 5 years old and may contain outdated information.
Science & Tech

By preserving delicate structures and tissues unlikely to fossilize under normal circumstances, deposits such as British Columbia’s Burgess Shale provide a startling record of the rapid diversification of early complex life — the so-called Cambrian Explosion. How this occurred has remained enigmatic. Bob Gaines, a geology professor from California’s Pomona College, however, believes he has unlocked the mystery.
Gaines and an international team collected physical and chemical evidence from the Burgess Shale and six similar-aged deposits in China and North America, pegging their extraordinary preservation to severe restriction of microbial activity after burial, due to a lack of oxygen and sulfate normally respired by microbes during decomposition. Furthermore, the mechanism of this restriction appeared to be successive formation of thin carpets of calcium carbonate on the sea floor — mineral barriers that prevented exchange of seawater with fossil-bearing sediments below. With the Burgess Shale itself, formed below the geologic structure known as the Cathedral Escarpment, each critter-entombing mud flow off the scarp was followed by formation of a corresponding carbonate layer. But why, researchers wondered, were Cambrian seas characterized by high calcium carbonate and low oxygen and sulfate levels?
Here, geochemical forensics led researchers to the Great Unconformity, a long-acknowledged global geologic phenomenon in which igneous and metamorphic basement rocks of the Precambrian are overlain by a several-kilometre thick layer of Cambrian sediment. While life flowered in the oceans but had yet to occupy land, these continental rocks underwent unprecedented erosion. Gaines and company hypothesized that the accompanying high rates of chemical weathering would have profoundly impacted ocean chemistry.
As falling rain mixes with atmospheric CO2 it becomes more acidic, leaching elements from rock and transporting them in dissolved form. It’s an important part of the geologic carbon cycle: during chemical weathering, atmospheric CO2 is transformed to bicarbonate (HCO3–) and delivered to the oceans, combining there with calcium (Ca2+) to produce calcium carbonate (CaCO3). Many marine organisms — both micro- and macroscopic — use this compound to produce mineralized shells and skeletons that are subsequently fossilized.
Thus, high concentrations of calcium carbonate may not only have restricted the decomposition of organic matter that led to Burgess Shale-type preservation, but helped spur the Cambrian Explosion by promoting early acquisition of skeletons.
Are you passionate about Canadian geography?
You can support Canadian Geographic in 3 ways:

Science & Tech
From Roberta Bondar to Harriet Brooks, Canada has more than its fair share of women scientists to be proud of. However women are still a minority in the STEM fields

Science & Tech
Researchers at the Royal Ontario Museum offer first glimpse of a spineless, thumb-sized animal based on Cambrian fossils found in the Rocky Mountains

Science & Tech
As geotracking technology on our smartphones becomes ever more sophisticated, we’re just beginning to grasps its capabilities (and possible pitfalls)

Environment
Carbon capture is big business, but its challenges fly in the face of the need to lower emissions. Can we square the circle on this technological Wild West?